Regarding monoclonal antibody access initiatives for LMICs: A pragmatic and straightforward opinion
By James W. Kim
Edited by Charles O. Johnson
Executive Summary
To maximize the impact of developing monoclonal antibody therapeutics against infectious diseases in and for LIC/LMIC environments, we must separate the effort to develop products which have an almost certain fate of commercial failure for the foreseeable future, from the effort to invest and develop a viable production infrastructure that can ultimately be utilized by the product pipeline to drastically de-risk product development and thereby significantly increase the chances of commercial success. This effort, along with precision-targeted funding to develop platform technologies enabling the leapfrog of mAb products to overcome typical LMIC infrastructure limitations must be the first steps taken before scarce resources are spent on developing actual products.
Background
The global monoclonal antibody (mAb) market has experienced exponential growth over the past two decades, becoming one of the most important classes of biopharmaceutical products. Yet access to these transformative therapies remains severely limited in low- and middle-income countries (LMICs), where the burden of infectious diseases is highest.
Multiple initiatives have been launched to address this gap, ranging from technology transfer programs to voluntary licensing agreements. However, despite these efforts, meaningful progress has been limited. The fundamental issue is not a lack of good intentions, but rather a misunderstanding of the structural barriers that prevent mAb products from being viable in LMIC markets.
What LMICs Have and Lack in the mAb Value Chain
LMICs generally possess a growing base of talented scientists and clinicians who are increasingly engaged in mAb research. Several countries have made significant investments in academic research infrastructure. What they critically lack, however, is the manufacturing infrastructure necessary to produce mAbs at scale and at a cost point that makes them accessible.
The production of monoclonal antibodies requires sophisticated biomanufacturing capabilities — from cell line development to large-scale bioreactor operations, downstream processing, and stringent quality control. These capabilities require not just capital investment, but deep operational expertise that takes years to develop.
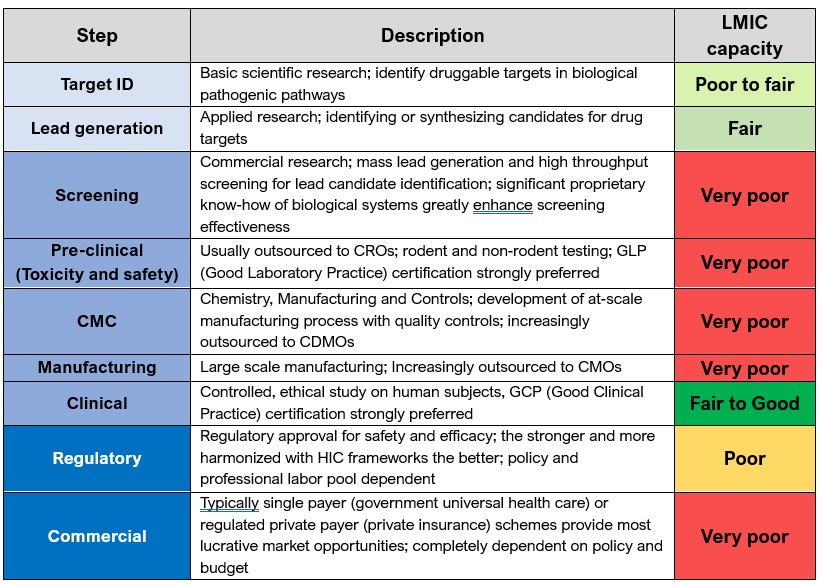 Overview of major steps in a modern mAb drug development pathway with assessment of LMIC capacity at each stage.
Overview of major steps in a modern mAb drug development pathway with assessment of LMIC capacity at each stage.
The State of mAb Therapies: The Nuance
It is important to understand that the mAb therapeutic landscape is far more nuanced than it appears at first glance. While blockbuster mAbs for oncology and autoimmune diseases generate tens of billions in revenue in high-income markets, the picture for infectious disease mAbs is fundamentally different.
Infectious disease mAb products face unique challenges: the patient populations are concentrated in settings with the least purchasing power, the diseases they target often have seasonal or epidemic patterns making demand unpredictable, and the existing standard of care — while suboptimal — is often extremely inexpensive.
The Truth About Viability in the Real World
The uncomfortable truth is that developing mAb products specifically for LMIC infectious disease indications is, under current conditions, commercially unviable. The cost of goods for mAb production — even at the most efficient facilities — remains orders of magnitude higher than what LMIC health systems can afford.
This is not a problem that can be solved by simply building a manufacturing facility. The entire ecosystem — from reliable cold chain infrastructure, to trained healthcare workers for administration, to pharmacovigilance systems, to functioning reimbursement mechanisms — must be in place for mAb products to reach patients.
Having Your Cake But Not Eating It Yet
The solution, we argue, lies in a sequential approach rather than trying to solve everything simultaneously:
Step 1: Build the Infrastructure First (The TSMC Model)
Just as Taiwan Semiconductor Manufacturing Company (TSMC) revolutionized the semiconductor industry by providing world-class manufacturing services to companies that could not afford to build their own fabs, the mAb space in LMICs needs dedicated Contract Development and Manufacturing Organizations (CDMOs).
These CDMOs would:
- Serve multiple product developers, achieving economies of scale
- Build deep operational expertise through continuous operations
- Attract and retain talent by offering stable, high-quality employment
- Generate revenue from contract manufacturing for high-income market products while building capability for LMIC products
Step 2: Invest in Platform Technologies
In parallel, targeted investment should go toward platform technologies that specifically address LMIC infrastructure limitations:
- Thermostable formulations that reduce cold chain dependence
- Subcutaneous or even oral delivery systems that eliminate the need for IV infusion infrastructure
- Rapid point-of-care diagnostic companion tests
- Simplified dosing regimens appropriate for community health worker administration
Step 3: Then Develop the Products
Only once manufacturing infrastructure exists and enabling technologies are available does it make sense to invest in developing mAb products for LMIC indications. At that point, the risk-reward calculus changes dramatically.
Closing Thoughts
All efforts must be made to plan and establish a viable, self-sufficient healthcare infrastructure. The stubborn focus on “getting things” to LMICs instead of fundamentally building up capability for LMICs to be independent, equal and ultimately more lucrative trading partners has meant that very little added value remained.
The mAb space presents a unique opportunity to get this right — but only if we resist the temptation to rush product development before the foundation is laid. Infrastructure first, platform technologies second, products third. This is not a popular sequence, because it delays the gratification of announcing new products for neglected diseases. But it is the only sequence that leads to sustainable, long-term access.
The pharmaceutical industry learned this lesson decades ago with small molecule generics: sustainable access came not from donations or voluntary licenses, but from building world-class manufacturing capability in India and China. The mAb revolution in LMICs will follow the same pattern — or it will not come at all.